icipe discovers trypanosome biomarkers and develops an inclusive diagnostic innovation for African animal trypanosomosis
Every year in Africa, three million cattle succumb to African animal trypanosomosis, a debilitating disease of livestock that is caused by trypanosome parasites, which are mainly transmitted by tsetse flies, and mechanically by other biting flies. Known as nagana in cattle, goats and sheep, and as surra in camels, the disease’s symptoms include anaemia, fever, swelling of the lymp nodes and decreased appetit; leading to weight loss, lethargy abortions, a suppressed immune system and increased chances of concurrent infections. The productivity of infected animals is reduced and thus, the availability of livestock products and services like tilling of agricultural land, while livestock production costs are escalated due to investments for treatment. African animal trypanosomosis causes direct and indirect losses of approximately USD 4.75 billion per year in Africa.
One of the main challenges in tackling African animal trypanosomosis is difficulties in its diagnosis. The main diagnostic strategies, which include microscopic examinations and polymerase chain reaction (PCR) tests, are technical, costly and often inaccessible to livestock keepers. Moreover, microscopy is not sensitive enough to detect asymptomatic trypanosomosis cases, where parasite loads are low. Often, livestock keepers make speculative, symptom-based diagnosis of African animal trypanosomosis. However, the disease’s signs are not unique to it, and they could result from other ailments. As a result, a high proportion of African animal trypanosomosis infections are either undetected or misdiagnosed. Lack of treatment or wrong therapies lead to livestock deaths, and in the latter case, drug resistance.
Now, researchers from the International Centre of Insect Physiology and Ecology (icipe) have developed a much needed non-invasive, rapid, affordable, efficient and easy method to diagnose African animal trypanosomosis. In a paper published recently in Frontiers in Microbiology journal, the icipe scientists report that they have identified biomarkers in the urine of cows, which indicate with certainty the presence of trypanosomes infection, even at low levels that would not be detectable through microscopy.
“Using this knowledge, we have developed a process for biomarker-based diagnosis of African animal trypanosomosis. As a result, through a simple urine test, which can be administered even by livestock keepers, it is possible to confirm whether an animal has the disease,” explains Dr Merid Getahun, icipe scientist and leader of the study. (Watch short video)
Knowledge to innovation
The icipe study investigated the mechanisms of the interactions between trypanosomes and their host animals. In general, a healthy animal produces a range of chemical compounds that may be used for a variety of functions, for example defence against disease causing agents. Once the trypanosome parasites enter the blood of an animal, a coevolutionary arms race starts between them and their host. The parasites fight to create a conducive environment for their own survival, for example by producing molecules that suppress the immune system of the host. In turn, the host produces antibodies and defence compounds to ward off the trypanosomes. In the process, the odour profile of the defence compounds in the host animals is changed.
“We identified two classes of compounds in the urine of cows, which are specifically affected by trypanosome infections. The first class of compounds are phenolics, which are ordinarily produced by the animals, but their production is elevated by the infections. The second class of compounds are ionones, whose production is specifically stimulated by the infections. Therefore, these two classes of compounds are predictive biomarkers of African animal trypanosomosis,” explains Dr Getahun.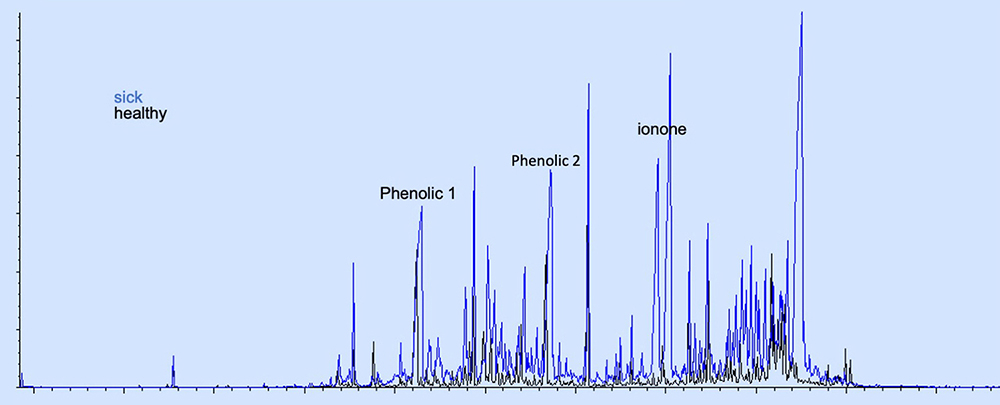
Image generated from icipe research showing the levels in phenolics and ionones in a healthy cow (black) and the changes of these compounds in a trypanosome-infected cow (blue).
Through laboratory and field studies, the researchers confirmed the potential of using the phenolic compounds for biomarker-based diagnosis of African animal trypanosomosis. The test involves injecting a reagent that is sensitive to the compounds into a urine sample. An activator is also applied to the solution. If the urine sample is from a healthy cow, it will remain green – the colour of the reagent. The colour of the urine sample from a trypanosomes infected animal will change to blue, indicating that the phenolics are above threshold level. The higher the parasite load, the darker the colour.

The images above show the results of the icipe biomarker-based diagnosis for African animal trypanosomosis against PCR and microscopy tests. (l-r): negative result on urine of a healthy cow based on PCR and microscopy tests; positive result on urine of a cow diagnosed as trypanosome positive using microscopy and PCR tests; positive result on the urine of an asymptomatic cow that diagnosed as trypansomosis positive through PCR tests, but negative through microscopy.
As shown in the photos above, the icipe biomarker-based diagnosis is highly sensitive and specific. It can detect a wide spectrum of trypanosomes infections, as well as the three most important trypanosome species. The test can also identify active forms of African animal trypanosomosis, as it returns a negative result for animals after treatment. Therefore, it can be used to monitor the effectiveness of therapeutic interventions. Moreover, the test is specific to trypanosome infections, as it is negative on animals suffering from other ailments with symptoms similar to African animal trypanosomosis.
“Supported by the right partnerships and pathways, the biomarker-based diagnosis can be translated into a ready-to-use, inclusive innovation. For example, the reagent and activator can be packaged into a dipstick that can be easily applied to a urine sample. This would empower livestock keepers to make evidence-based decision on the health of their animals,” says Dr Dan Masiga, Head, icipe Animal Health Theme.
Transformative potential
“This simple, novel diagnostic tool for African animal trypanosomosis is emblematic of icipe’s mission of developing science-led, inclusive innovations that are effective, accessible and affordable to all segments of society, and which have a transformative impact on communities and economies,” states Dr Segenet Kelemu, icipe Director General & CEO.
The technology becomes part of icipe’s arsenal to fight the tsetse fly menace, towards an overall one health goal, through sustainable livestock farming to improve food security, household incomes and nutrition, while addressing the challenges of climate change and protecting the environment.
Notes for Editors
Research details: Getahun MN, Ngiela J, Makwatta JO, Ahuya P, Simon TK, Kamau SK, Torto B, Masiga D.(2022) Metabolites from trypanosome-infected cattle as sensitive biomarkers for animal trypanosomosis. Frontiers in Microbiology. doi: 10.3389/fmicb.2022.922760
Collaborating institutions: International Centre of Insect Physiology and Ecology (icipe), and Kabete Veterinary Laboratories, Nairobi, Kenya.
Corresponding authors: Merid Getahun (MGetahun@icipe.org)
Funding: This study was supported by the German Ministry for Economic Cooperation and Development (BMZ) through the Deutsche Gesellschaft für Internationale Zusammenarbeit (GIZ) BMZ 81219442 project number: 16.7860.6-001.00; BMZ/GIZ ICTDL grant no. 18.7860.2-001.00; Centre for International Migration and Development (CIM) of GIZ and the German Federal Employment Agency; Max Planck Institute support to the icipe Partner Group; European Union, through the Integrated Biological Control Applied Research Program (IBCARP) grant no. DCIFOOD/ 2014/346-739 – 608; Norwegian Agency for Development through the Combating Arthropod Pests for Better Health, Food and Resilience to Climate Change (Norad-CAP-Africa) RAF-3058 KEN-18/0005. We gratefully acknowledge the icipe core donors: Swedish International Development Cooperation Agency (Sida); Swiss Agency for Development and Cooperation (SDC), Federal Democratic Republic of Ethiopia and Government of the Republic of Kenya. The views expressed herein do not necessarily reflect the official opinion of the donors.
The International Centre of Insect Physiology and Ecology (www.icipe.org): Our mission is to help alleviate poverty, ensure food security, and improve the overall health status of peoples of the tropics, by developing and disseminating management tools and strategies for harmful and useful arthropods, while preserving the natural resource base through research and capacity building.
African animal trypanosomosis is caused by trypanosome protozoan parasites, the most important being Trypanosoma congolense, T. vivax and, to a lesser extent, T. brucei group. The trypanosomes are transmitted by the Glossina tsetse fly species robust, sparsely bristled insects that usually range from 6 to 16 mm (0.2 to 0.6 inch) in length, which are only found in Africa, especially in the woodlands and grasslands. Trypanosomes are also spread mechanically by other vectors, for example horse flies and stable flies, which obtain the parasites when they feed on infected animals and then transfer them to other animals. Due to this mode of transmission, coupled with the movement of livestock, African animal trypansomosis is also present outside tsetse infested areas. It is estimated that 50 million cattle and 70 million sheep and goats are at risk of African animal trypanosomiasis, in Africa, Asia and Latin America.

The icipe tsetse fly management arsenal includes: the NGU trap (left) and the tsetse repellent collar (right).

